PersistenceMarketResearch says that the global Phenylketonuria Treatment Market will witness a CAGR of more than 11% between 2020 and 2030.
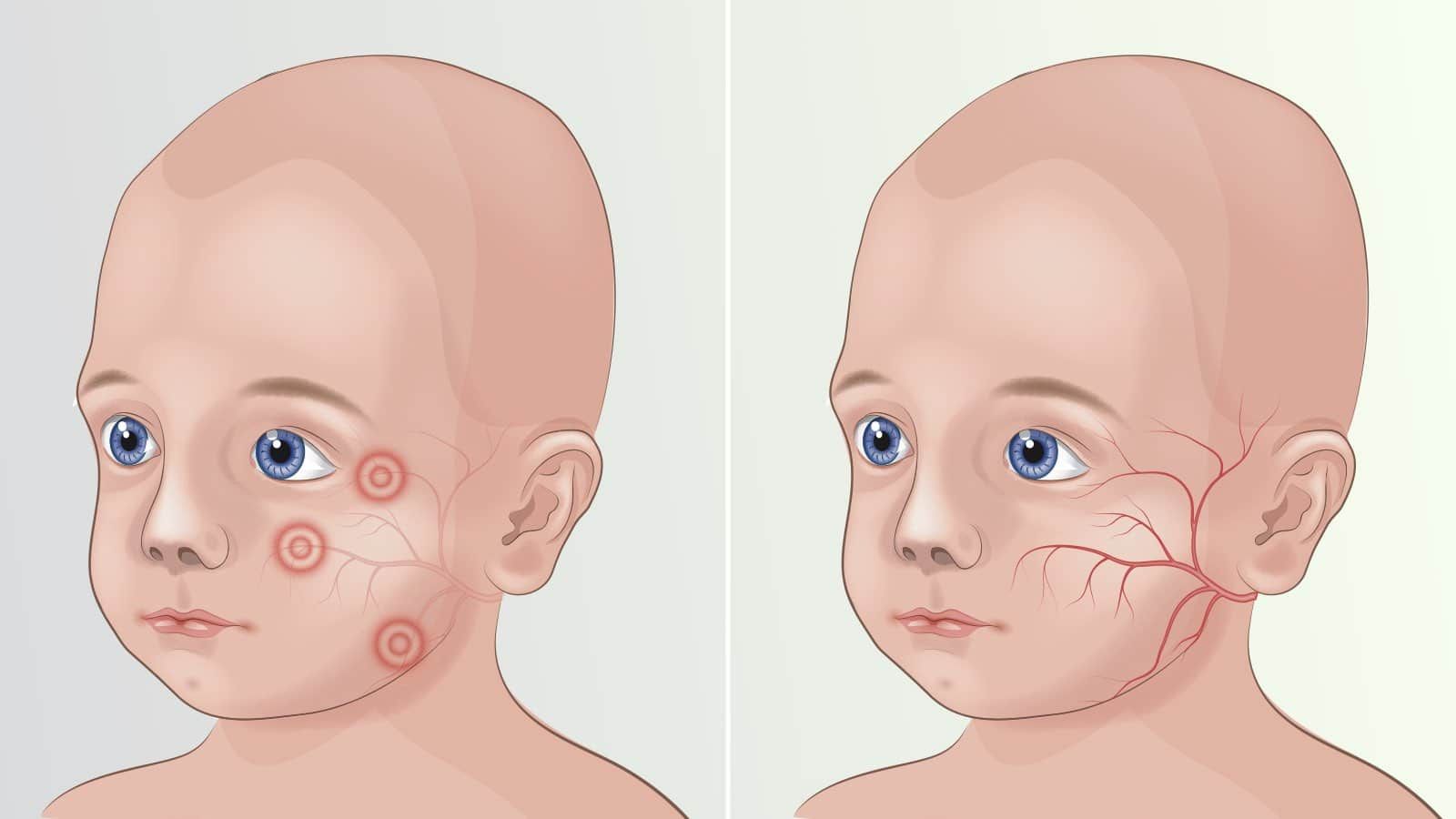
Incidences of Phenylketonuria have been rising at an exorbitant rate since the last few years. As per the Genetics Home Reference, 1 out of 10K-15K cases of Phenylketonuria is seen in new-borns all across the US. The certain European countries like Ireland, 1 out of 4500 new-borns contract Phenylketonuria, whereas in Turkey, the figure is 1 in 4000 individuals.
The pipeline drugs like CNSA-001, RTX-134, and SYNB1618 are expected to rejuvenate the Phenylketonuria treatment market in the years to come. The agencies like the US FDA, EMA (European Medicines Agency) are also on the verge of fast-track approval herein. Along these lines, after enzyme therapy drug- Palynziq (pegvaliase) being launched by BioMarin Pharmaceuticals, Inc for treating Phenylketonuria in the US in 2018 got approved by the MAA (marketing Authorization Application) and EMA in the same year. The drug was launched in May 2019 worldwide.
Get the detail info about the article: https://www.persistencemarketresearch.com/market-research/phenylketonuria-treatment-market.asp
North America dominates the Phenylketonuria treatment market. This could be attributed to feasible regulations coupled with increase in number of initiatives by the governments for treating Phenylketonuria. Europe stands second. The Asia-Pacific is expected to pick up pace between 2020 and 2030 with advancement in awareness regarding Phenylketonuria.
The other market participants include Censa Pharmaceutical, Inc., Rubius Therapeutics, Inc., Synlogic, Inc., Retrophin, Inc., and Homology Medicines, Inc. On the developments’ front, Retrophin, Inc., in May 2019, did provide funds at the initial stage of Rare Caregiver Respite Program. NORD (National Organization for Rare Disorders) had, of late, launched the program with the objective of providing numerous services and monetary assistance to patients. Also, Homology Medicines Inc., has come up with HMI-102, a gene therapy for Phenylketonuria. The IND (Investigational New Drug) application on its part has already received approval from the US FDA.