As per PersistenceMarketResearch, the global Atrial Fibrillation Devices market will witness a remarkable CAGR between 2020 and 2030.
Market Snapshot
Hypertension and valvular heart disease are among the common alterable risk factors of atrial fibrillation. The atrial fibrillation devices are emerging as alternatives to the oral anti-coagulants that have been used for a long time. At the same time, drug therapy is also being explored as an alternative treatment for atrial fibrillation, which could act as a restraint on the atrial fibrillation devices market. For individuals seeking expert advice, consulting a vascular doctor in Singapore can provide additional insights into managing these risk factors and exploring treatment options.
Going Deeper
Out of diagnostic and catheter ablation devices, catheter ablation devices are holding a larger market share due to increasing demand for the non-pharmacological treatments. On the technological developments’ front, the Kardia Mobile device comes across as one of the portable smartphone compatible accessories containing 2 electrodes. The device does transmit signal to Kardia Mobile app. Therein, recording begins with placement of left and right fingers on 2 electrodes. As such, ECG recording gets activated and the report flashes on smartphone’s screen. Approved by the US FDA, it has 98% specificity and 97% sensitivity for detection of Atrial Fibrillation. Likewise, there are other devices in queue to see the light of the day.
Know More about Atrial Fibrillation Devices Market: https://www.persistencemarketresearch.com/samples/31531
How about Region-wise?
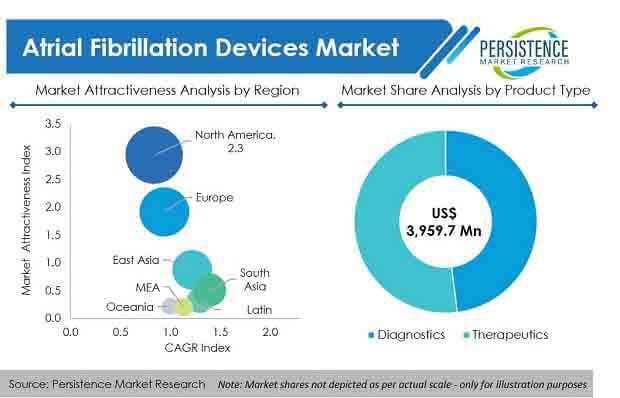
North America holds the largest market share. This could be attributed to the fact that the US is the primary adopter of new-fangled technology. Europe comes in second due to Germany witnessing an ever-increasing application on the part of minimally-invasive catheter ablation procedures. As far as the Asia-Pacific is concerned, China is expected to rule the roost due to increasing consumption of tobacco amongst the teenagers. Plus, the number of baby boomers is rising year after year. LATAM and MEA are also expected to catch up going forward.
Competitive Landscape The key market participants are inclusive of Siemens AG, Microport Scientific Corporation, Johnson & Johnson, Abbott Laboratories, Boston Scientific Corporation, St. Jude Medical, Inc., and Medtronic Plc., Bioscience Webster, and AtriCure, Inc.